Will 30% of Early Stage (1-IIIA) go on to metastasize??
Comments
-
sasamat, thanks for your input and for verifying that my thought process makes sense (at least a bit!).
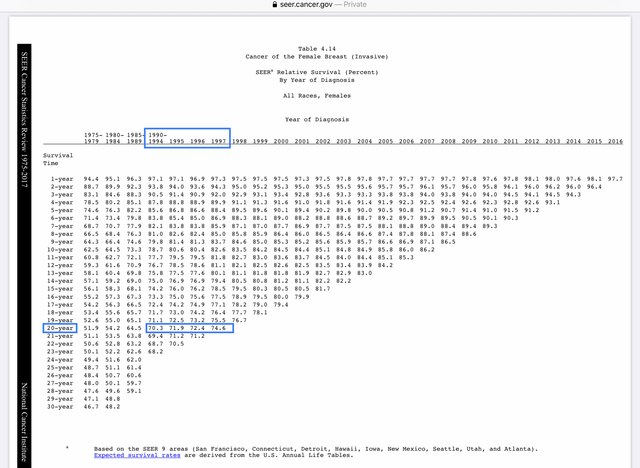
moth, take a look at the 20 year survival stats from the SEER data. Note that the most recent 20 year data that is available is for those diagnosed in 1997. If you look at the 4 periods that I estimated to be within the range of the study you linked, you'll see the following 20 year survival rates:
1990-1994: 70.3% (75.3%)
1995: 71.9% (76.9%)
1996: 72.4% (77.4%)
1997: 74.6% (79.6%)
The SEER data includes all stages. Stage IV de novo consistently represents 5%-6% of all diagnoses. So the number in brackets represents the 20 year survival rate for all breast cancer patients diagnosed in that year, when you exclude the 5% who were Stage IV de novo. That doesn't quite match "early stage", since it includes Stages IIIA and IIIB. But just looking at these figures, what you can see is that if 30% of early stagers were to eventually develop mets (i.e. if the long term survival rate for early stagers is 70%), this means that even in 1990-1994, 5% of early stage patients who would eventually succumb were surviving longer than 20 years. By 1997, when the 20 year mortality rate was 79.6%, it means that almost 10% of those patients were still alive at 20 years. This is why I assumed that those who passed away between 2008 and 2012 were diagnosed as much as 20 years earlier. If anything, I was conservative in that estimate and should have assumed that those who passed away in 2008 might have been diagnosed as much as 30 years earlier.
moth, to your comment, "Because tamoxifen and herceptin came on board just before 2000 so those benefits should be seen in those earlier 2008-12 numbers, shouldn't they?", the answer is No.
Herceptin was approved for metastatic breast cancer in 1998. It was approved for node-positive non-metastatic cancer in 2006 and it was approved for early stage node negative after that. The same is true of AIs - they were barely in use and I believe not approved for early stage back when I was diagnosed in 2005. Even Tamoxifen was considered the new wonder drug for early stagers back in 2005 - it was approved but as I recall not all insurance companies paid for it yet. So if most of those who passed away between 2008 and 2012 were initially diagnosed in the early 2000s or prior to 2000, most would not have had access to any of these drugs. The other significant change would be 3rd generation chemos. So in fact there have been hugely significant treatment changes between the group discussed in the study you linked, and those diagnosed today.
Edited to correct the spelling of sasamat's name. Sorry for the error!
0 -
Lilly,
May I ask you how did you find your 3 mm lobular tumor? I mean which device was able to catch it? Did your biopsy take the whole tumor?
0 -
I posted this elsewhere so I know Beesie already saw it :-) but this article might be a valuable contribution to this thread. I personally find the 30% statistic to be plausible when considering lifetime metastatic recurrence risk.
The article (which is recent) states that "[a]lthough the incidence of distant relapse has been shown to be decreasing and survival times for patients with recurrent disease have improved, 20–30% of patients with early breast cancer still die of metastatic disease."
This article talks about many interesting things. One of which is that a very complicating factor in counting lifetime metastatic recurrence risk is that many HR+ cancers recur decades after initial diagnosis. DECADES! Personally, I would have loved to have decades before my HR+ metastatic recurrence, but that's another story. Hopefully for all those out there who recur later, better treatments and cures will be found by then.
Anyway, the article is an excellent read for those who want to delve into the why and how of metastatic recurrence. There is still a lot we do not know.
0 -
Beesie,
Could you please help me with some positive statistics regarding young women (30-40) and AI treatment? I read results from one study about benefit of the ovarian suppression+AI to prevent cancer reoccurrence. But they also mentioned that this treatment did not increase overall survival rate because of increased mortality from other issues. I assume heart problems were the major cause. I want to find something more optimistic.
0 -
Beesie and Sasamat thank you for the excellent information. To Moth's point, there is such a need to collect, evaluate, and disseminate data in fighting this disease.
Buttonsmarch - that is an interesting article. Thank you for posting.
0 -
buttonsmachine, yes, that is a very interesting article.
However to the quote "20–30% of patients with early breast cancer still die of metastatic disease.", here is what I replied in the other thread in which buttonsmachine posted this same information *** I suspect that the 20% figure is probably fairly accurate, if not low - but 20% is 33% less than 30%, which to me is a huge difference. Based on the number of people diagnosed every year with early stage breast cancer in North America, over 10 years the difference between 20% and 30% would be an additional 250,000 people who survive and never develop mets. And note that while the article is recent, the sources for the "between 20% and 30%" stat are from 2000 and 2005, which means that current treatments are not reflected in these figures.***
And for additional clarification, my discussion here (and elsewhere) about the 30% stat is talking about lifetime metastatic risk, not the 5 or 10 or 15 year risk that we see reflected in the Oncotype score and the PREDICT and CancerMath models. Note the SEER chart that I posted above, which shows 30 year mortality rates. Of course, you can only have 30 year mortality figures for those who were diagnosed more than 30 years ago. So to sasamat's point, there is simply no way to know the lifetime probability of distant recurrence for someone diagnosed today.
What we do know, as buttonsmachine points out, is that some recurrences don't happen until decades after the initial diagnosis. Take a look here (this is a chart that I have posted previously in this thread). It shows recurrence patterns/timelines for each major subtype of breast cancer.
Source: Factors that predict recurrence later than 5 years after initial treatment in operable breast cancer https://wjso.biomedcentral.com/articles/10.1186/s12957-016-0988-0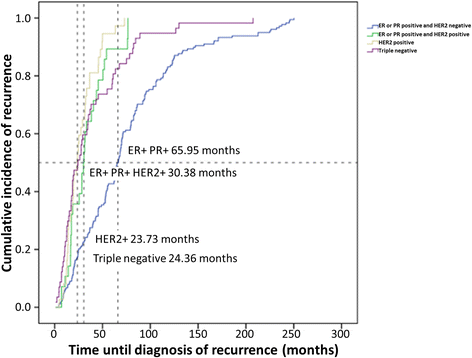
It appears that almost all recurrences for HER2 positive disease happen within the first 6 years. 80% of recurrences of TN disease happen within 5 years. ER+/HER2- disease is the one that tends to have a larger percent of late recurrences, but even here, it appears that about 80% of recurrences happen within the first 10 years, and about 95% occur within 20 years. This information helps frame this discussion about lifetime recurrence risk. If we can get a handle on 10 year or 15 year recurrence rates (as the Oncotype and PREDICT and CancerMath models tell us), we can estimate lifetime recurrence risk from there.
All that said, we have to remember that these figures are nothing more than estimates, even when we are looking at recurrence risks based on our own diagnoses, as determined by Oncotype and PREDICT and CancerMath. As for the much disputed 30% stat (which I believe today would be closer to 20%), which is a generalized stat that's been calculated to reflect outcomes based on an average of hundreds of thousands of people with every possible "early stage" diagnosis, well, that is absolutely meaningless for any one individual. And that, I suppose, is the point of all my posts and why I hate it so much whenever this 30% stat is brought up again. Too often, that stat is misinterpreted to reflect individual risk, and in that context, it is misleading and irrelevant and too often creates unnecessary fear and angst.
MikaMika, I'm sorry but I can't help you on your question. It's not something that I have ever read up on.
Edited to correct typos (I was in a bit of a rush this morning).
0 -
Beesie, thank you as always. My feeling is that MO takes into account other conditions like in my case probably since I am PR-, small tumor that has spread to nodes.
Mika, 3mm was discovered after BMX biopsy. 2 positive nodes were found at that time as well.
0 -
Here are a couple of very simple stats to consider:
In 2020, an estimated 276,480 new cases of invasive breast cancer are expected to be diagnosed in women in the U.S., along with 48,530 new cases of non-invasive (in situ) breast cancer.
About 42,170 women in the U.S. are expected to die in 2020 from breast cancer. Death rates have been steady in women under 50 since 2007, but have continued to drop in women over 50. The overall death rate from breast cancer decreased by 1.3% per year from 2013 to 2017. These decreases are thought to be the result of treatment advances and earlier detection through screening.
Just to use a blunt instrument to estimate risk, 42170/276480 = 15.25%.Yes, the vast majority of those who died were first diagnosed in a different year. How long ago? Ten years ago or less? Probably. Has the rate of new diagnoses changed a lot? Not much, about .3% a year, according to this.
- Breast cancer incidence rates in the U.S. began decreasing in the year 2000, after increasing for the previous two decades. They dropped by 7% from 2002 to 2003 alone. One theory is that this decrease was partially due to the reduced use of hormone replacement therapy (HRT) by women after the results of a large study called the Women's Health Initiative were published in 2002. These results suggested a connection between HRT and increased breast cancer risk. In recent years, incidence rates have increased slightly by 0.3% per year.
So maybe the risk of death is somewhere between 15-20% across all invasive cases, from early to de novo diagnosis.
Stats in this comment are from this link. https://www.breastcancer.org/symptoms/understand_b...
0 -
'28% of deceased from breast cancer were early stage', isn't equal to 28% of early stagers go on to have a metastatic recurrence, is it? Aren't they comparing percentages from two different populations?
0 -
My dad's best friend is a very notable oncologist. I didn't get to use him since I live in another state but he helped me a lot through my treatment. When the BCI test came out he was very quick to tell me to have it done because most studies showed an accuracy rate far more than 99.9%. At the time I had it done (I came back high risk of recurrence, albeit the very low end, but low benefit from continuing Tamoxifen which is very uncommon.) he told me that the oncology community across the board feels the advances made in breast cancer treatment in the last 5 years are equal to the advances made in the 40 years before that. He also told me that most of them think they'll have a "cure" for breast cancer in my lifetime (I'm 57) so fingers crossed. And when I was first diagnosed he told me that I would probably outlive my peers because due to my cancer, I would receive much better medical care than most women with my physical stats. It's been quite a delight to pick his brain.
0 -
lala, when I was diagnosed stage 1 in Dec 2017, my mother's very experienced physician told her I'd just have a sucky year of surgery and treatment and live to a ripe old age.
Now I'm stage 4 and odds are my mother will outlive me /sigh/
0 -
Moth, I know how you feel. A month before my diagnose I was told by my GP that I am so healthy that I will be a healthy very old woman. How ironic, isn't it?
With trillions of dollars poured for a cancer cure, it has become more of a profit than a cure. We are going from stage I to IV and we are losing our sisters every day because there is no cure. I'm sorry, I am feeling down anytime I hear we will get cancer cure in our lifetime.
0 -
Lilly,
Thank you for your answer. I just thought that some hospitals have very sensitive imaging equipment.
0 -
Very interesting discussion. One comment, as one of those who had “decades" between original diagnosis and recurrence I'm not sure if I feel particularly lucky. The reality is, numbers aside, BC is unfortunately a disease that hangs over us until we die of something else. I have a friend that used to describe it as the Sword of Damocles. In my case, while I was early stage, there were many things that pointed to the outcome I am now experiencing. And yet, at almost 16 years out, I was at the point that I was no longer looking over my shoulder. 🙄
All to say, generalized numbers are not helpful. As Beesie has so eloquently stated, the stats are hopelessly outdated and then the uniqueness of our individual diseases muddy the waters. I do feel that, for the majority of patients things go well. For some of us, not so much - crap shoot. 😉
Edited to correct a bizarre iPad typo.
0 -
Thank you everyone for your thoughtful discussion. I have been away from the forum for a bit and things in the world are making my heart heavy. My Oct 2020 annual mamm has been delayed due to covid and I feel a mounting sense of panic growing inside. I fear the mamm. I now know that a blissful life going in one direction as if I didn't have a care in the world can change on a dime. With one bad mamm. I fear the mamm. I fear not having one. I don't want to have my annual mamm. I don't want to miss it. Do I sound crazy? I am.
I fought taking tamoxifen tooth and nail. I struggled with it. I resented it. It does seriously crappy things to me. Killer leg cramps and vicious hot flashes at the top of the list. Seized joints and a vagina that feels like a bag of dry popcorn are next on the list. Yet the day my onc tells me I don't have to take it anymore, I think I'm going to feel shoved off a cliff. What will be between me and that 15, 20 or 30% recurrence coming my way? How stupid is that? Hate taking that pill and then not wanting to stop taking it.
I think we all wish there was some way we could feel safe. Okay. Girded with some solid knowledge to hang onto. But there is no such thing. We are not safe. Oh god, I am sucking so bad today! I am sorry this thread gives you angst Beesie, but I do have it in my faves because this issue turns over and over in my mind. Especially when I find a new lump (buried somewhere in all the fat, wait, no, it's a cookie I lost earlier, never mind).0 -
runor, I think most of us can identify with what you wrote. And to be fair, my angst over this thread is really just angst over the title of this thread. The discussion is interesting and important. The problem is that too many people read the title, read the first thread, believe the 30% to be fact, and spread it beyond this thread. The 30% stat has taken on a life of it's own.
The title of the thread was written in October 2013. The opening post was last edited in November 2013. It was a valid question at that time and the 30% was likely accurate at that time, recognizing that long-term survival stats by their nature are always based on people diagnosed a long time ago. So in reality the 30% stat was probably never accurate for those newly diagnosed at the time of reading. Regardless, a lot has happened since 2013. That's all been discussed in the 68 pages of posts but I doubt that anyone new to this thread is reading 68 pages of posts. They read the opening post and maybe the latest few posts. That's why I have to keep re-posting the same information over and over again, which doesn't give me angst but does tick me off.
Today, in 2021, the title of this thread is fear-mongering click-bait.
I wish there was some way to change the title to something more reflective of the current discussion, such as "What % of Early Stage (IA - IIIA) go on to metastasize?". Then I would be just fine with this thread.
0 -
Can mods not alter the title of the thread??
runor, I thought you were near me? do you have a date for your mammo? Cause if not, I know places with appts. There were supposed to be no delays for existing cancer pts. BC is pretty committed to no delays for us (surgical, imaging or treatment) hmu by pm if u want deets
0 -
Beesie, "That's all been discussed in the 68 pages of posts but I doubt that anyone new to this thread is reading 68 pages of posts."
I think I did actually read all 68 pages!
0 -
Yes, I too did read all 68 pages. Those of us who are interested will... But likely if Susan's Garden hadn't titled the thread as she did (30%) I never would have opened it in the first place. There are so many thread started that one has to be a bit selective in choosing what to read. So in the end, I'd hate for the title to be changed. edited to say - (Sorry Beesie)
0 -
MountainMia, I'm impressed!

I've been posting since the early days of the thread so I've read all of it but I've never gone back more than a few pages to review the discussion. I should do that. It would offer an interesting longer-term perspective.
0 -
I have been following this thread since I joined this community, but I admit I haven’t read all the pages.
Even though the title may not be 100% accurate it appears to be pretty close and the discussions have been very interesting, probably one of the most intersecting threads i have seen over the past six plus years.
I am not sure the name needs any change, after all what would materially change if it were 20 years vs 30? The bottom line is still the same, i.e. a very large number will progress sooner or later.I personally feel at peace with this projection, but I admit it took a certain adjustment to get there psychologically.
Peace and health to all
0 -
I'm sorry I can't stop laughing at Runor's cookie
 )
) 0
0 -
Beesie - The writer in me objects to any changes without author permission. However, I think the mods could certainly add an edit to the original post that tells people not to freak out (as if by the time you get to BCO you haven't been massively freaking out already!) and directs them to perhaps the last 20 pages of posts where a more accurate depiction of stats can be found. That would be fair in my mind. Not a change, an addendum.
Moth - I am in the Interior. Shuswap area. I do not yet even have an appointment. When I called the booking dept they said they were backed up. I said I was already 2 months past my mamm date. The lady said they would be calling me in 5 or 6 months. This is not cool. I'm really thinking I should call my doc and screech a little. But then I think, is my boob situation any worse than anyone else's boob situation? Should I bump some other terrified woman who is also waiting? What's so special about me that I should get preferential treatment? My mind is not a quiet nor simple place.
Lilly - nothing better than finding free food in the fat folds!
0 -
runor...I think that idea for the moderators to do would be a heavy, complicated task. When i was diagnosed 11 years ago, I came here because there was so little info on the net about my rare type of BC. That said, what little info I could find was posted on the mucinous bc thread. That thread was a lifeline for me because I was able to find small nuggets of info that allowed me to pursue other info out there on the net.
I have kept the mucinous thread growing as new internet travellers have found their way to it and even some of them, with the info THEY have found BECAUSE of it are able to fine tune the thread because of what THEY found or EXPERIENCED.
Would I be comfortable if the mods added an addendum to the mucinous thread? No.
BUT...what the mods could do is explain SOMEWHERE ...when looking at older threads, readers should be careful in trying to understand that very long threads, started years ago, may not contain current info.
But...that should be obvious to a reader. Readers need to be critical thinkers. If not, they need to find an advocate who can help them CRITICALLY understand info.
Readers also need to understand statistics and if they don't understand it, they need to take a crash course in it OR again, find an advocate who does understand statistics.
And finally, when SusanGardens posted this thread, after I read it and wet my pants, I showered, changed into my armour gear and set out to find out whether or not the statement was affirmative.
Eleven years later, I haven't progressed YET... so....do I still wonder IF and/or I will? Sure I do. A very sobering thought and feeling.
However, in those same 11 years I have had other health scares that usurped my worrying about a BC recurrence. A few years ago, through imaging for a hip ailment, the radiologist found a 5 cm tumor inside my thigh that needed to be removed. So, I thought to myself...VR, here you were worrying about a breast cancer recurrence and this stupid tumor in your leg might kill you instead...
The damn tumor was benign and the replacement hip is wonderful.
So...I no longer worry about bc recurring. I don't care about statistics either any more. They are too fluid for my taste or experience.
As the DH, an engineer, will always say to VR when she finds a study about ANYTHING and is gently walking her back from the plank...Dear, it is ONLY one study...wait a day, there will be an other...
Stay safe.
Stay well!
And most of all...stay SANE.
0 -
runor, I said "I wish there was some way to change the title to something more reflective of the current discussion". I very intentionally worded that as a wish and not a request or expectation. I know that the Mods don't change subject lines without author permission, and in this case, the author has not posted in two years or even signed on to BCO in 20 months.
muska is right in that nothing materially changes if the number is 20% or 30%. The fact is that for each of us as individuals, nothing would change even if the number was 10%. The 30% figure, or whatever the correct figure currently is, has no bearing on any of us as individuals. If we've had invasive breast cancer, we face our own individualized risk of metastatic breast cancer. For some of us, that risk is very low, maybe only 5%. For others, that risk is much higher, maybe 40%. So the 30% (or whatever) is meaningless, except for medical planners and researchers. That's why I don't understand the constant dredging up of this thread, or the obsession with the 30% and with proving the 30% to be correct. My obsession with the 30% is based on the fact that I think the subject line is fear-mongering click-bait. I spend a lot of time on this board writing posts to try to calm people down by providing rational, clear, hopefully accurate information. The subject line here does the exact opposite.
0 -
Bessie. Can I know your initial diagnosis. It is missing in signature.
I follow all your post and it is helpful
0 -
I took statistics at the college level. The first thing our professor told us was this saying:
"There are lies, damned lies, and statistics."
This was drilled into us over and over throughout the course. Statistics gives us sets of raw data, the problem is with the interpretation of the data. It is not as straightforward as some people assume it is. Hence the saying.
Anyway, it's important to remember that statistics, or medical science, rarely has the final answer to anything. It's all a moving target and a work in progress. I am comfortable with taking all statistics and projections with a grain of salt.
0 -
Beesie, while i appreciate that many newbies need to be talked down from panic and reassured, I'm actually disturbed by how many women go through treatment for early stage and have NO CLUE about recurrence risk or that stage 4 is terminal. Literally have had women say they didn't know it could come back, because the drs. don't use the word 'remission' anymore, but rather they're told they're cured. Many don't know what symptoms to monitor for when looking for recurrence and again, some have no idea that stage 4 is fatal and has a pretty short survival time.
Also, knowing that there is a population risk of recurrence and that metastatic recurrence is fatal is IMO critical to moving 'breast cancer awareness' campaigns beyond the pom poms and "I survived!" cheerleading. We stage 4 people have limited time and energy to mobilize for effective treatments that actually prolong life. We need our early stage peers to take that up on our behalf and I think one reason they're not is that they have no idea of what the risks are.
So there's a time & place for what I think is a 'reality check' on this disease, and recognition that early stagers can go on be be one of the 41,000 people in US who die of breast cancer each year & that we're still not very good at predicting who will and who won't.
0 -
Moth, I 100 agree with everything you said and have said on this thread. You said it much kinder than I was going to say it.
I was told by my first oncologist that I'd be CURED. Whoops. This thread actually is what got me to research recurrence and metastatic risk when I was poking around the forums early stage. Nobody told me anything I read here. Doctors, nurses, therapists. Everyone was too busy talking me down. Maybe if everyone had been honest and ditched the pom poms and things had been explained to me in the beginning I would of done better coping with my early stage diagnosis and my soon thereafter metastatic recurrence. Thats what makes me grind my teeth. Lack of information and transparency. Too many women as moth explained dont understand things they should. I feel ultimately I was only semi prepared for what happened because of my own advocacy.
0 -
I appreciate the fact that statistics for the entire population and one’s own individual risk are very different. In fact, one way I retained hope that I might not die in two years after my Stage IV diagnosis was by hanging on to Stephen Jay Gould’s story and particularly this quote: The median is not the message.
With that said, I support the point that moth is making. In the first few weeks before my mets were discovered I was told by my BS that my (supposed) Stage 2 IDC was “very curable”. My mets were only discovered because a breast MRI (only breast, we weren’t looking for metastasis) showed something that looked like a lung nodule so I was referred for a PET scan, and the rest is history. My “curable” Stage 2 was really Stage 4.
My MO confirmed that I would not have gotten any imaging of the rest of my body had it not been for the suspected lung nodule. I would have been treated as a “curable” stage 2 and after a year I would have gone off Herceptin. I had NO symptoms of any kind other than my breast lump. Yet I had moderately widespread bone Mets.
That caused me to think about how easily mets can be missed and I read on Komen’s site about the 30% stat. In trying to avoid “futile” imaging how many cases are missed? I don’t think giving everyone a PET at diagnosis is feasible but what moth says about monitoring for symptoms (and making sure our doctors take them seriously) is important
0